Comparing LAMA with LABA and LTRA as add-on therapies in primary care asthma management | npj Primary Care Respiratory Medicine
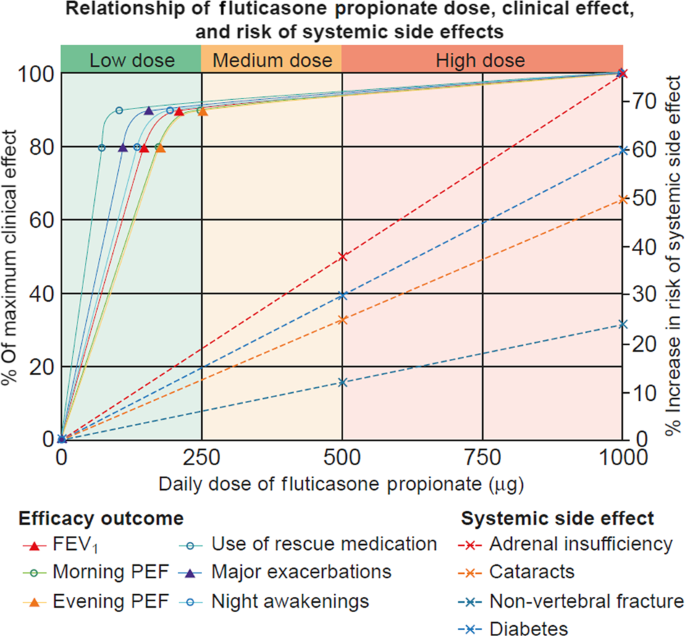
Comparing LAMA with LABA and LTRA as add-on therapies in primary care asthma management | npj Primary Care Respiratory Medicine

Petition · Legal requirement for strong warning about risk of Singulair and generic Montelukast · Change.org

Intermittent montelukast in children aged 10 months to 5 years with wheeze (WAIT trial): a multicentre, randomised, placebo-controlled trial - The Lancet Respiratory Medicine

Neuropsychiatric side reactions of leukotriene receptor antagonist, antihistamine, and inhaled corticosteroid: A real-world analysis of the Food and Drug Administration (FDA) Adverse Event Reporting System (FAERS) - World Allergy Organization Journal