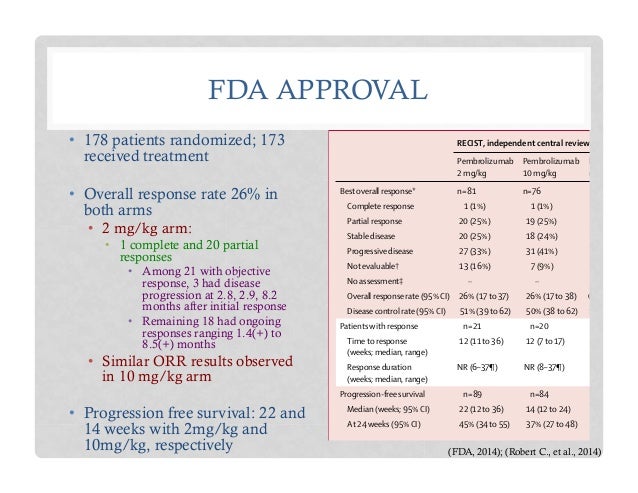
FDA Grants Accelerated Approval to Pembrolizumab in Combination with Pemetrexed and Carboplatin for Treatment of Metastatic Non-Squamous NSCLC | ONS Voice
Cancer Immunotherapy Update: FDA-Approved Checkpoint Inhibitors and Companion Diagnostics | SpringerLink

FDA Approval Summary: Pembrolizumab plus Lenvatinib for Endometrial Carcinoma, a Collaborative International Review under Project Orbis | Clinical Cancer Research

A reality check of the accelerated approval of immune-checkpoint inhibitors | Nature Reviews Clinical Oncology

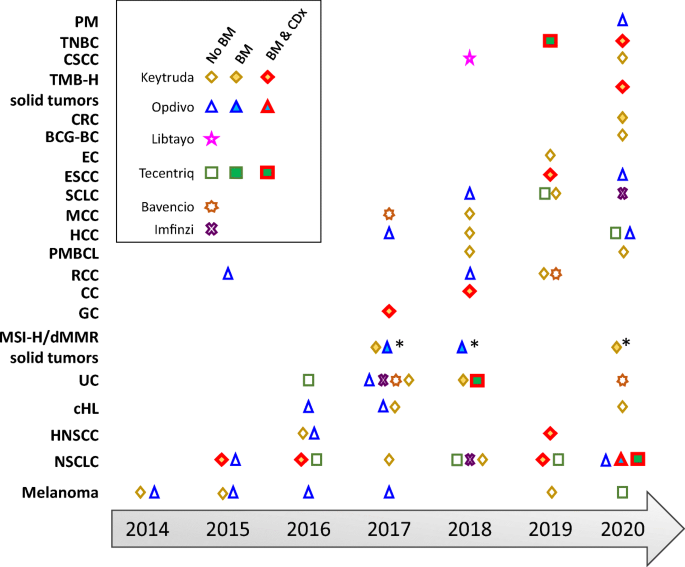
Time line of FDA approvals for the anti-PD-1 antibodies pembrolizumab... | Download Scientific Diagram
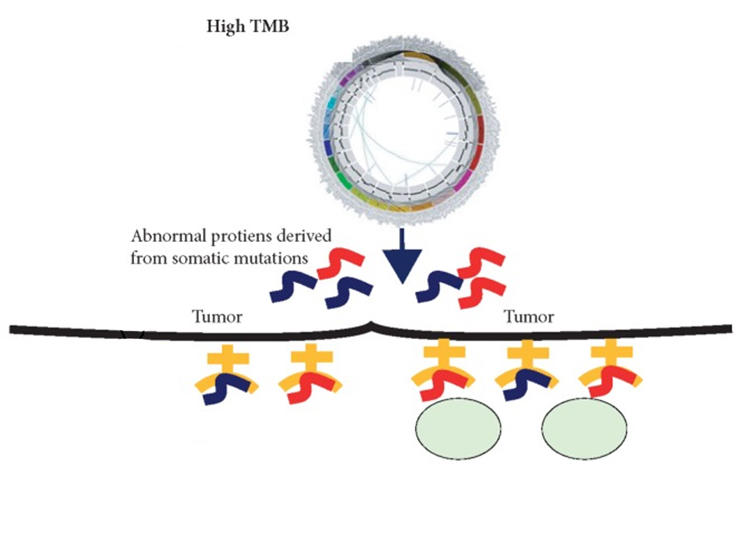
![PDF] FDA Approval Summary: Pembrolizumab for the Treatment of Microsatellite Instability-High Solid Tumors | Semantic Scholar PDF] FDA Approval Summary: Pembrolizumab for the Treatment of Microsatellite Instability-High Solid Tumors | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/d87ced43628213babff1c81f9a45fcf02a22d940/2-Figure1-1.png)
PDF] FDA Approval Summary: Pembrolizumab for the Treatment of Microsatellite Instability-High Solid Tumors | Semantic Scholar

Merck's Keytruda (pembrolizumab) Receives the US FDA's Approval for Label Update to Treat Advanced Urothelial Carcinoma
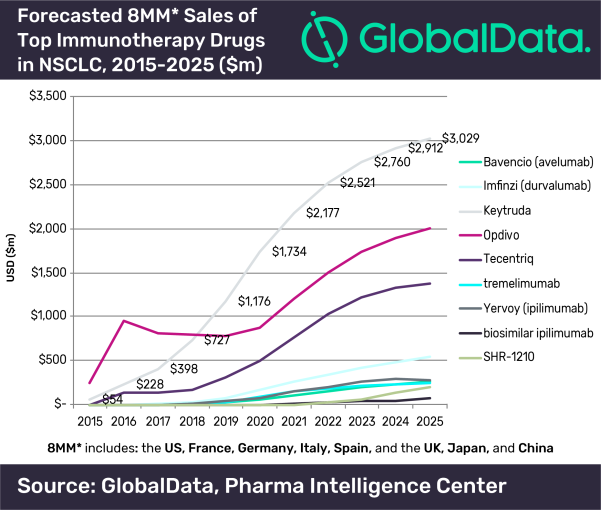
Approval of Keytruda in Europe for first-line use with chemotherapy in lung cancer strengthens the use of PD-1 inhibitors as backbone therapy, says GlobalData - GlobalData